ACER Stock Drops on the Failure of Phase II Study. Cash Updates
Acer Therapeutics ACER released top-line results of its phase IIa proof-of-concept study to evaluate ACER-801, (osanetant), as a treatment option for moderate to severe Vasomotor Symptoms. ACER-801, the study candidate, failed to achieve statistical significance for its ability to reduce hot flashes severity or frequency in postmenopausal females. The drug was safe and well tolerated. On Friday, the stock fell 57%.
In the past year, the shares of Acer Therapeutics have dived 75.7% compared with the industry’s decline of 18.2%.
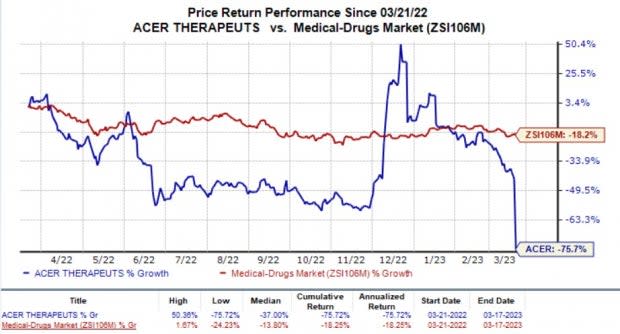
Image Source: Zacks Investment Research
ACER-801 (an investigational, non-hormonal neurokinin-3 receptor antagonist (NK3R), is currently under review for possible treatment of patients with VMS and prostate cancer. The company signed an exclusive license agreement in January 2018 with Sanofi SNY. Per the agreement, Acer acquired worldwide rights to osanetant from Sanofi, to develop osanetant as a potential treatment for certain neuroendocrine-related disorders.
Phase IIa of ACER-801 was conducted in 49 women between 40 and 65 who suffered from moderate to severe hot flushes. Over a 14-day period, the patient population was divided into four groups. Each cohort received either 50, 100, or 200 mg of ACER-801 twice daily. Safety and pharmacokinetics were the primary endpoints. The secondary endpoint evaluated efficacy in comparison to placebo treatment. After the disappointing results were announced, ACER-801 was halted and the company plans to complete an analysis of all data.
Acer already has Olpruva (sodium Phenylbutyrate), an approved drug in their portfolio. This drug is for the treatment urea cycles disorders involving deficiencies of carbamylphosphate synetase or ornithine trigarbamylase or arginasuccinic acid synthetase. In December 2017, Olpruva was approved for the indicated indication by the FDA. In June 2022, the company initiated a phase III DISCOVER Study of Edviso (celiprolol), to treat patients with COL3A1-positive vascular Ehlers-Danlos Syndrome.
Acer stated in the same press release that cash runways are running low for the company’s operations. The company’s cash and cash equivalents will be sufficient to cover its capital and operating requirements until the second quarter in 2023. Acer will require additional financing in order for Olpruva to be launched in the U.S. However, Acer did not provide any assurance about the availability of capital at the required time and on acceptable terms. Acer may have to suspend, restructure or stop product development, commercialization, or other operations if it does not receive the funds. Acer’s stock price plunged significantly on Friday due to this.
Acer Therapeutics Inc. Price & Consensus
Acer Therapeutics Inc. price-consensus-chart | Acer Therapeutics Inc. Quote
Zacks Rank and Stocks To Consider
Acer Therapeutics is currently ranked #3 on Zacks (Hold).
These stocks are worth looking at Avalo Therapeutics AVTX and Aziyo Biologics AZYO and AZYO both have a Zacks Rank 2 (Buy) currently. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
In the last 90 days, Avalo’s loss estimates for the year 2023 have narrowed from $3.62 to $2.57 per share. Avalo’s shares plunged 72.5% over the past year.
AVTX exceeded estimates in two quarters reported, with an average earnings surprise (37.83%)
In the last 90 days, Aziyo’s loss estimates for the year 2023 have narrowed from $2.14 to $1.65 per share. Aziyo’s shares have dropped 9.5% over the past year.
AZYO beat estimates in four of the most recent quarters but missed the mark on three other occasions. The average loss surprise was 15.24%.
Get the most recent Zacks Investment Research recommendations. Download 7 Best Stocks in the Next 30 Days Today Click to get this free report
Sanofi (SNY) : Free Stock Analysis Report
Acer Therapeutics Inc. (ACER) : Free Stock Analysis Report
Aziyo Biologics, Inc. (AZYO) : Free Stock Analysis Report
Avalo Therapeutics, Inc. (AVTX) : Free Stock Analysis Report